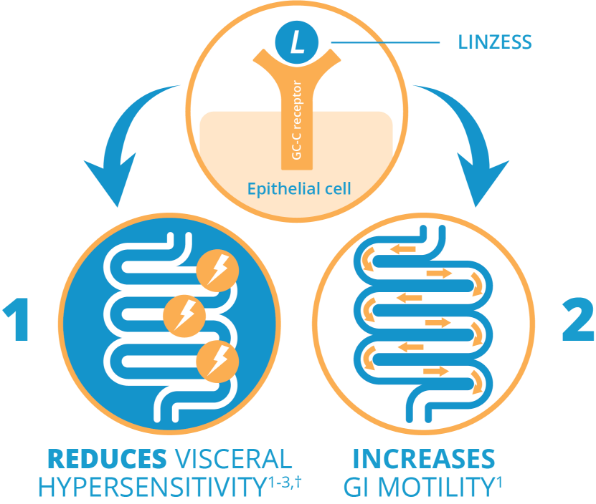
INDICATIONS AND USAGE
LINZESS® (linaclotide) is indicated for the treatment of:
- irritable bowel syndrome with constipation (IBS-C) in adults
- chronic idiopathic constipation (CIC) in adults
- functional constipation (FC) in pediatric patients 6 to 17 years of age
IMPORTANT SAFETY INFORMATION
WARNING:
RISK OF SERIOUS DEHYDRATION IN PEDIATRIC PATIENTS LESS THAN 2 YEARS OF AGE LINZESS is contraindicated in patients less than 2 years of age; in nonclinical studies in neonatal mice, administration of a single, clinically relevant adult oral dose of linaclotide caused deaths due to dehydration.
Contraindications
- LINZESS is contraindicated in patients less than 2 years of age due to the risk of serious dehydration.
- LINZESS is contraindicated in patients with known or suspected mechanical gastrointestinal obstruction.
Warnings and Precautions
Risk of Serious Dehydration in Pediatric Patients Less Than 2 Years of Age
- LINZESS is contraindicated in patients less than 2 years of age. In neonatal mice, linaclotide increased fluid secretion as a consequence of age-dependent elevated guanylate cyclase (GC-C) agonism, which was associated with increased mortality within the first 24 hours due to dehydration. There was no age-dependent trend in GC-C intestinal expression in a clinical study of children 2 to less than 18 years of age; however, there are insufficient data available on GC-C intestinal expression in children less than 2 years of age to assess the risk of developing diarrhea and its potentially serious consequences in these patients.
Diarrhea
- In adults, diarrhea was the most common adverse reaction in LINZESS-treated patients in the pooled IBS-C and CIC double-blind placebo-controlled trials. The incidence of diarrhea was similar in the IBS-C and CIC populations. Severe diarrhea was reported in 2% of 145 mcg and 290 mcg LINZESS-treated patients and in <1% of 72 mcg LINZESS-treated CIC patients. In pediatric patients 6 to 17 years of age, diarrhea was the most common adverse reaction in 72 mcg LINZESS-treated patients in the FC double-blind placebo-controlled trial. Severe diarrhea was reported in one LINZESS-treated patient. If severe diarrhea occurs, dosing should be suspended and the patient rehydrated.
Common Adverse Reactions (incidence ≥2% and greater than placebo)
- In IBS-C or CIC adult patients: diarrhea, abdominal pain, flatulence, and abdominal distension.
- In FC pediatric patients: diarrhea.
Please see full Prescribing Information, including Boxed Warning, or visit https://www.rxabbvie.com/pdf/linzess_pi.pdf.
US-LIN-230215
References: 1. LINZESS. Prescribing information. Allergan, Inc.; 2021. 2. Feng B, Kiyatkin ME, La JH, et al. Activation of guanylate cyclase-C attenuates stretch responses and sensitization of mouse colorectal afferents. J Neurosci. 2013;33(23):9831-9839. doi:10.1523/JNEUROSCI.5114-12.2013 3. Eutamene H, Bradesi S, Larauche M, et al. Guanylate cyclase C-mediated antinociceptive effects of linaclotide in rodent models of visceral pain. Neurogastroenterol Motil. 2010;22(3):312-e84. doi:10.1111/j.1365-2982.2009.01385.x